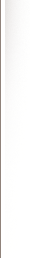I woke up this morning with Fusion and Fission on my mind. I must have had some interesting dreams or trips during the night.
So I wanted to create an article dealing with the current view of fusion, fission, transmutation, and the cycle of the elements. I wanted to approach this from a layman, as I am, and to try and piece this together with a 2-way Universe. Current understanding is that this is a 1-way, expanding Universe, and only 1 end of the cycle is really taken into consideration.
There are 2 ends to the pump, or 2 ends to the cycle. Newton had it right that the apple fell from the tree, but how did the apple get up there in the first place?
We need to add in the growing, living end of the cycle. So before I go into my thoughts and try to link this all together. Lets go over some definitions. These are mostly taken from Wiki.
Definitions:
Fusion: is a nuclear reaction in which two or more atomic nuclei collide at a very high speed and join to form a new type of atomic nucleus. During this process, matter is not conserved because some of the mass of the fusing nuclei is converted to photons (energy).
Fission: is either a nuclear reaction or a radioactive decay process in which the nucleus of a particle splits into smaller parts (lighter nuclei).
Transmutation: is the conversion of one chemical element or isotope into another. In other words, atoms of one element can be changed into atoms of another element by ‘transmutation’. This occurs either through nuclear reactions (in which an outside particle reacts with a nucleus), or through radioactive decay (where no outside particle is needed). —-has been proven by multiple experiments starting with Rutherford.
Radioactive: See decay below
Radioactive Decay: is the process by which a nucleus of an unstable atom loses energy by emitting particles of ionizing radiation. A material that spontaneously emits this kind of radiation—which includes the emission of energetic alpha particles, beta particles, and gamma rays—is considered RADIOACTIVE. -same as fission to me
Alpha decay: Alpha decay occurs when the nucleus ejects an alpha particle (helium nucleus). Alpha particle – stated as being identical to a helium nucleus
Beta decay: Beta decay occurs when the nucleus emits an electron or positron and a type of neutrino, in a process that changes a proton to a neutron or the other way around. The nucleus may capture an orbiting electron, converting a proton into a neutron (electron capture). All of these processes result in nuclear transmutation. Beta particle – high-energy, high-speed electrons or positrons emitted by certain types of radioactive nuclei.
Gamma decay: are produced alongside other forms of radiation such as alpha or beta, and are produced after the other types of decay occur. Was almost always found associated with other types of decay, and occurred at about the same time, or afterward. Gamma ray – refers to electromagnetic radiation of extremely high frequency and therefore high energy per photon. (light bullets per Russell)
Ok… now lets start with one quote from Wiki that will help tie all this together.
The fusion of two nuclei with lower masses than iron (which, along with nickel, has the largest binding energy per nucleon) generally releases energy, while the fusion of nuclei heavier than iron absorbs energy. The opposite is true for the reverse process, nuclear fission. This means that fusion generally occurs for lighter elements only, and likewise, that fission normally occurs only for heavier elements. – Quote from Wiki
So fusion happens with lighter elements (before Carbon) and fission happens with heavier elements (after carbon). My take on the elements(inspired by the Russells) are seen to start with long slow nebulous waves which create lighter gaseous elements(before Carbon), then after carbon(balance point) speed up and shorten their waves creating an increase in density and sometimes mass. So there would be multiple octaves or levels of elements before Hydrogen. These not being isotopes… that would change everything. Considering we label Hydrogen as 1 and base everything else off that.
Also it states that fusion is possible for light and heavy…so here we see a hint at the starting point or growth cycle. Because of the growth and death cycles on each side of Carbon it differs on whether energy is released or absorbed. My take on this is that during the growth or compressing cycle before Carbon, energy can be released and reused in aiding to help in this compression cycle. After Carbon during the natural death, expanding, or centrifugal cycle, for fusion to happen it is required to absorb energy from the surrounding space to have the strength to complete the reaction. This is because it is naturally releasing energy.
If we look at the cycles having 2 streams… and each stream is fighting or resisting each other throughout the whole process, this will help in understanding. During the growth stage the compressing centripetal stream is growing in strength and ratio to the resisting centrifugal expanding stream. This friction causes heat too.
But in these cycles there will always be both. Just a preponderance of 1 or the other. So once it gets to Carbon or the maturation point, it switches and the dying decaying centrifugal stream takes over and natural fission begins to happen. Hence the description of radioactive elements.
Back again to the above statement…. fusion generally occurs for lighter elements and fission generally occurs with heaver elements. Fits right in if we look at 2 sides of the coin. Both can happen on each side but it requires a deviation from the norm and a change in energy being absorbed or released.
This can bring us into natural transmutation.
It is described as:
‘Natural transmutation is responsible for the creation of all the chemical elements we observe naturally. One type of natural transmutation observable in the present occurs when certain radioactive elements present in nature spontaneously decay by a process that causes transmutation, such as alpha or beta decay. An example is the natural decay of potassium-40 to the argon-40 which forms most of the argon in air. Also on Earth, natural transmutations from the different mechanism of natural nuclear reactions occur, due to cosmic ray bombardment of elements’
So here it states we have transmutation happening all around us at all different times. But they always focus on the decay or the radioactive process and forget to add the generoactive or living end. Transmutation can happen at any time in the cycle for any element. Besides, all elements are just different conditions of the same substance. Which can be manipulated at will by affecting fields and pressures.
The snippet about potassium decaying to argon is important too. Most elements will decay naturally to their inert gas. The inert gas is the origin of the element in that particular octave line. But it can go more in depth and decay all the way back to Helium and before if the reaction is strong enough. Here is a post to check out with some more inert gas info.
http://lightcoalition.org/cosmic-clock-inert-gases

I next want to talk about Fusion. There is not a whole lot mentioned about Fusion beyond the tritium-deuterium reactions which they say is the Sun’s fusion process of combining Hydrogen into Helium. I have been seeing more and more in the Scientific community about fusion and creating it in the lab, but they are still using Hydrogen(tritium and deuterium) and trying to implode or compress and fuse into Helium.
Fusion in the Sun:
‘It is the reaction in which two atoms of hydrogen combine together, or fuse, to form an atom of helium. In the process some of the mass of the hydrogen is converted into energy. The easiest fusion reaction to make happen is combining deuterium (or ‘heavy hydrogen) with tritium (or ‘heavy-heavy hydrogen’) to make helium and a neutron.’
I have a feeling this will be found later to be a process with different details. Once they find more elements below Hydrogen they will realize that this process is more likely to be happening even farther below in other octaves all combining together. I also think they will find more Carbon and that Carbon has more importance in the cycle.
It just doesn’t add up yet for me yet. That they would create Helium. Especially if Helium is found to have a different configuration than what they think(article coming soon).
So as I stated earlier, there are elements below Hydrogen that haven’t been found yet. Well lets just say labeled, because I think many of the properties and descriptions now given to H, are more likely to be assigned to separate elements earlier in the chart/process/cycle.
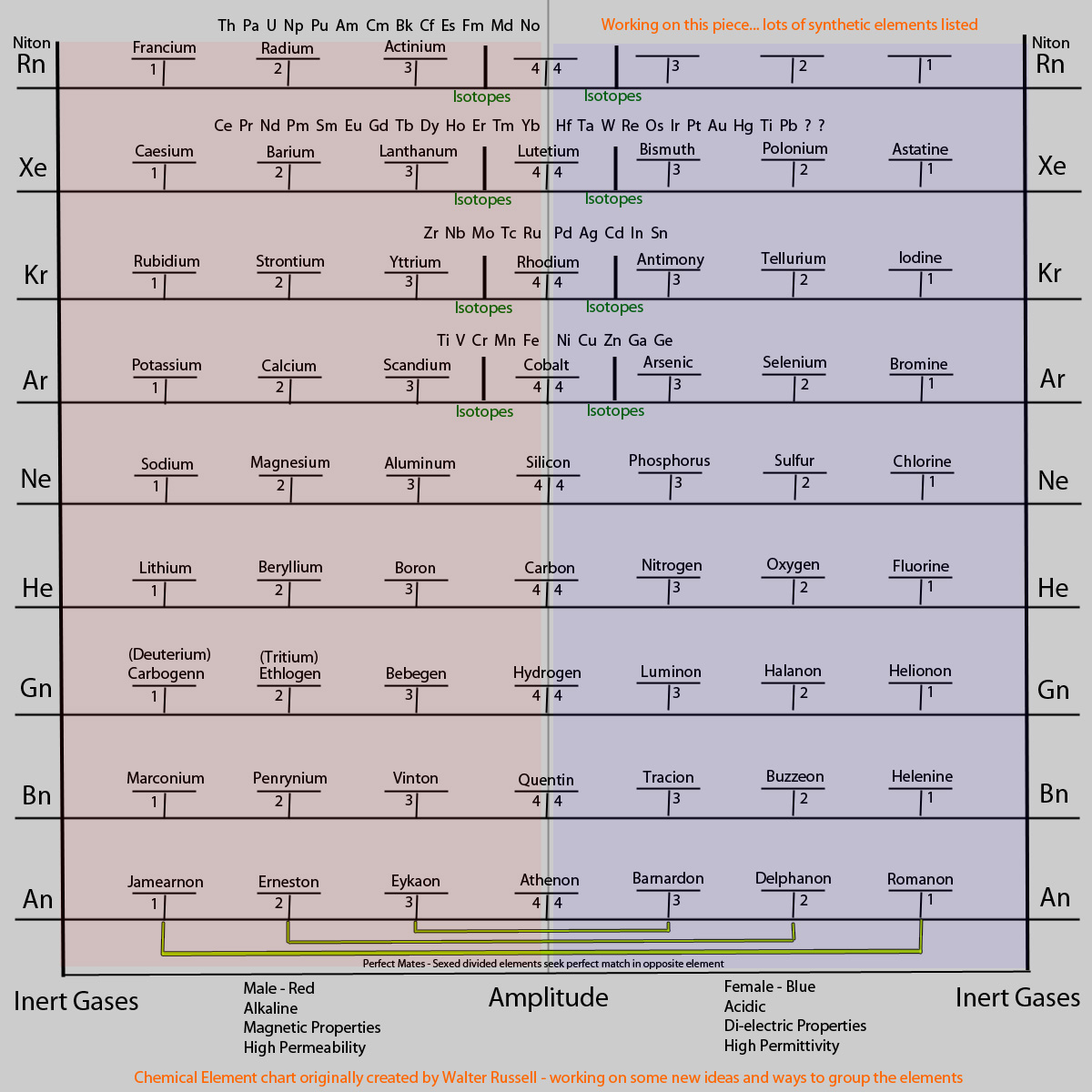
Tritium and Deuterium are said to be isotopes of hydrogen… yet they were found by Walter Russell to be elements in themselves.
Tritium for example can be created in reactions with Deuterium by absorption of ‘particles’. More transmutation…see?
In closing, this topic could go on for days, but I just wanted to get people thinking.
Here are some thoughts and questions.
Could all the elements be considered radioactive? I would say so…considering they all have both cycles. Maybe use different names.
What is the opposite of radioactive? So what would we call them? Stable or Generoactive / compressing / living?
There is a death and decaying cycle. So shouldn’t they have a growth or living cycle with all of them as well? Yup!
Would it not match this type of thinking?
Fusion – generoactive, compressing, living, charging, centripetal
Fission – radioactive, expanding, dying, discharging, centrifugal
There has to a growth cycle before you get to the radioactive elements, this is that what the lower lighter octaves are. Maybe Fusion is going up to carbon and fission going down to elements like Uranium.
Thanks for fighting through the urge to say… ugh this is too much! Thanks for taking this ride with me and much love to you all!
Chris